Cutting-Edge Software
MINEQL+ 5.0: Advanced Tools for Aqueous Chemical Equilibrium
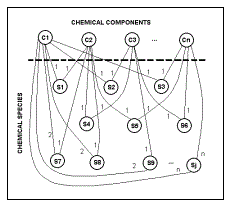
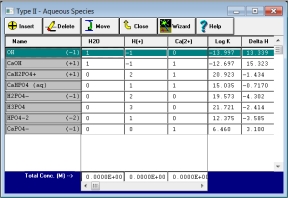
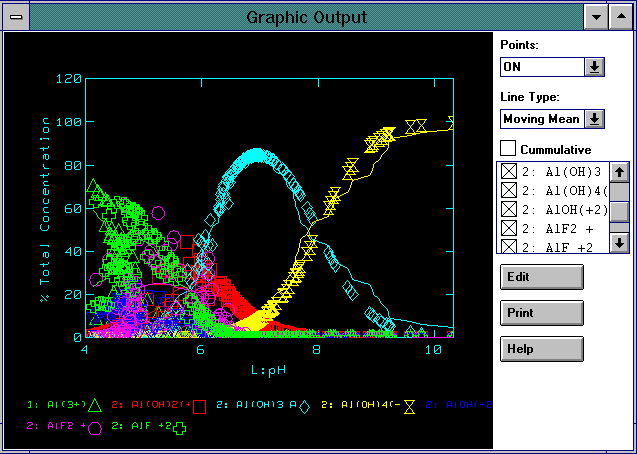

Professional Software Now Available for Free Download.
✓ 30 years of use and development
✓ Used in over 700 colleges and universities worldwide
Cutting-Edge Software




✓ 30 years of use and development
✓ Used in over 700 colleges and universities worldwide